Melting Temperature Of Metals Chart
Melting Temperature Of Metals Chart - Melting is the change of state from a solid to a liquid. Melting of a pure substance occurs at a particular constant temperature called melting point. Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. The state of being melted. The reverse processes are freezing and deposition,. Melting is the conversion of a solid to a liquid. In a pure crystalline solid, this process occurs at a fixed temperature called the melting point. The molecules in a solid, which are bound. Melting occurs when a substance absorbs heat energy, causing its particles to vibrate more rapidly until they break free from their fixed positions in the solid state. The temperature at which something begins to melt is. In a pure crystalline solid, this process occurs at a fixed temperature called the melting point. Melting occurs when a substance absorbs heat energy, causing its particles to vibrate more rapidly until they break free from their fixed positions in the solid state. The molecules in a solid, which are bound. Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. Melting is the change of state from a solid to a liquid. This occurs when the internal energy of the solid increases, typically by the. The state of being melted. The temperature at which something begins to melt is. The act or operation of melting. Melting describes the change of a solid into a liquid when heat is applied. The state of being melted. The reverse processes are freezing and deposition,. Melting is the change of state from a solid to a liquid. The molecules in a solid, which are bound. Melting is the process of changing something from a solid into a liquid (like metal into liquid metal). Melting is a process that causes a substance to change from a solid to a liquid. When a solid is converted directly to a gas, the process is known as sublimation. Melting occurs when a substance absorbs heat energy, causing its particles to vibrate more rapidly until they break free from their fixed positions in the solid state. The molecules. Melting of a pure substance occurs at a particular constant temperature called melting point. The quantity melted at a single operation or in one period. The molecules in a solid, which are bound. When a solid is converted directly to a gas, the process is known as sublimation. Melting is also known as fusion, although this term has several meanings. In physics and chemistry, melting is the process of converting a solid substance to its liquid form, typically by heating the substance to a temperature called its melting point. Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. In a pure crystalline solid, this process occurs at. Melting describes the change of a solid into a liquid when heat is applied. Melting is the process of changing something from a solid into a liquid (like metal into liquid metal). Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. The quantity melted at a single. Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when a substance absorbs heat energy, causing its particles to vibrate more rapidly until they break free from their fixed positions in the solid state. Melting is the conversion of a solid to a liquid. Melting is the process by which a. Melting of a pure substance occurs at a particular constant temperature called melting point. Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. When a solid is converted directly to a gas, the process is known as sublimation. The reverse processes are freezing and deposition,. This occurs. Melting is a process that causes a substance to change from a solid to a liquid. Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. Melting is the conversion of a solid to a liquid. Melting is the process of changing something from a solid into a. Melting of a pure substance occurs at a particular constant temperature called melting point. Melting is also known as fusion, although this term has several meanings in. The state of being melted. The reverse processes are freezing and deposition,. In a pure crystalline solid, this process occurs at a fixed temperature called the melting point. The molecules in a solid, which are bound. It is the opposite of freezing. Melting is the conversion of a solid to a liquid. The reverse processes are freezing and deposition,. In physics and chemistry, melting is the process of converting a solid substance to its liquid form, typically by heating the substance to a temperature called its melting point. Melting occurs when the molecules of a solid speed up enough that the motion overcomes the. The act or operation of melting. Melting is a process that causes a substance to change from a solid to a liquid. The state of being melted. Melting is the change of state from a solid to a liquid. The reverse processes are freezing and deposition,. Melting describes the change of a solid into a liquid when heat is applied. In physics and chemistry, melting is the process of converting a solid substance to its liquid form, typically by heating the substance to a temperature called its melting point. The molecules in a solid, which are bound. Melting of a pure substance occurs at a particular constant temperature called melting point. Melting is also known as fusion, although this term has several meanings in. Melting, or fusion, is a physical process that results in the phase transition of a substance from a solid to a liquid. Melting is the process of changing something from a solid into a liquid (like metal into liquid metal). The quantity melted at a single operation or in one period. In a pure crystalline solid, this process occurs at a fixed temperature called the melting point. The temperature at which something begins to melt is.Metals Melting Temperatures » Jewelry Artists Network Metal working, Melting temperature
Consolidated Chart of Melting Points for Metalsmiths Metals and Their Solders
Metal Melting Point Chart Metals Silver
Metals and their Melting point ranges. Download Table
Melting Point Definition and List
Metals Melting Reference Chart
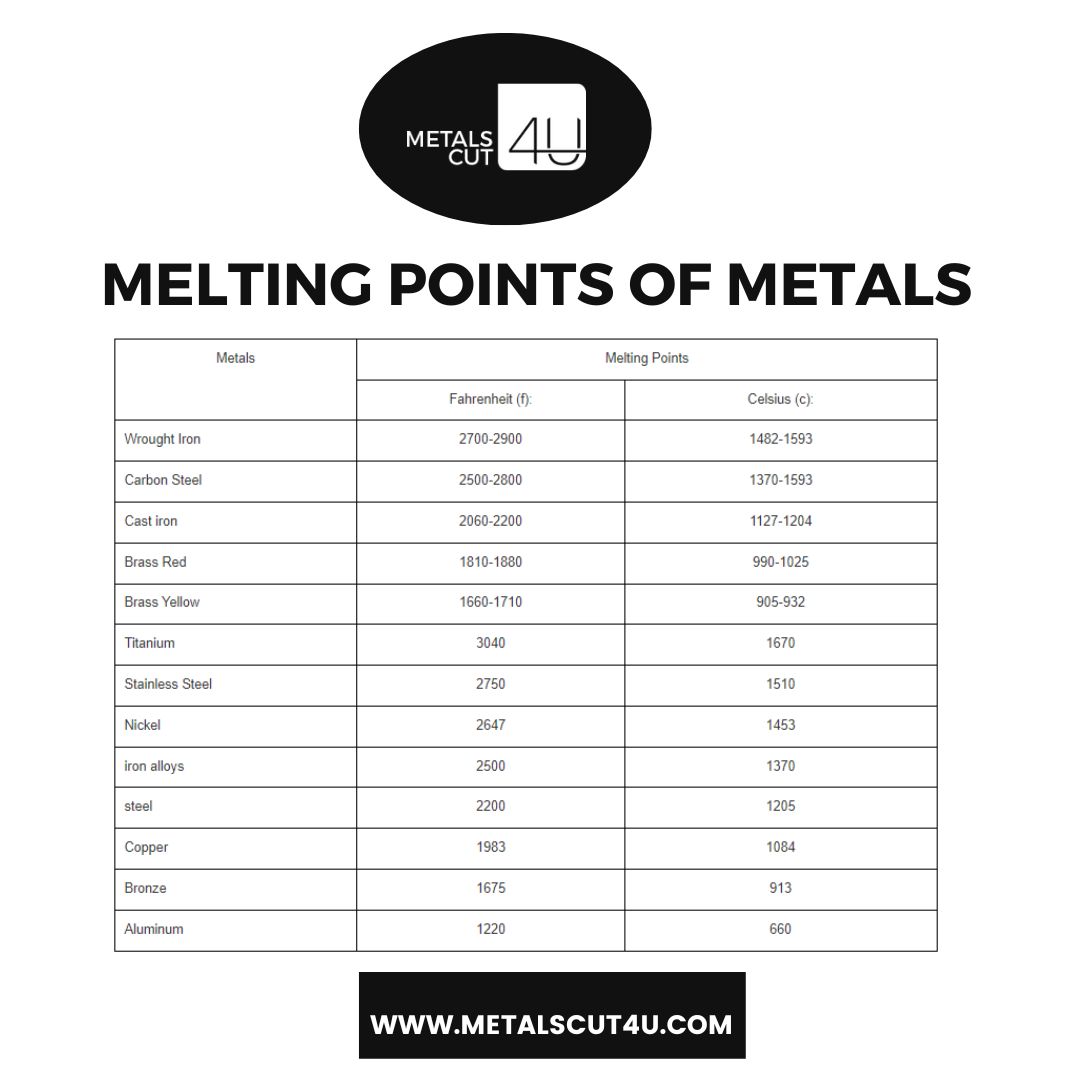
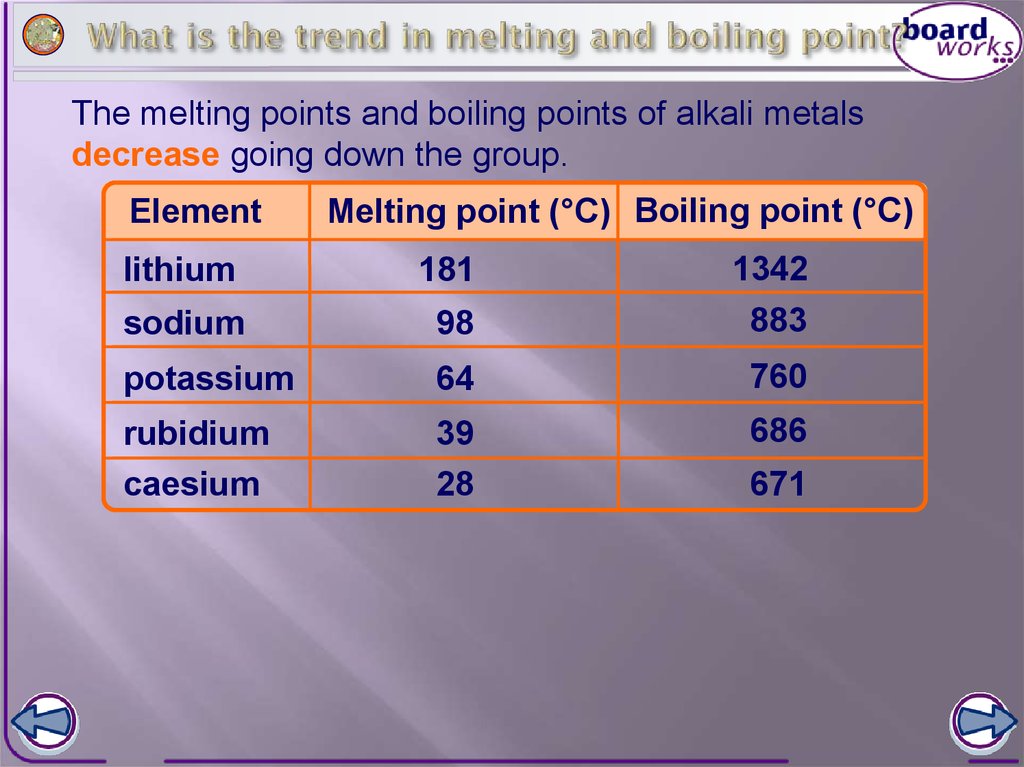
Melting Points of Metals
melting point of metals chart
melting point chart Melting metals highest points point why energy dissociation enthalpy bond
metal melting points chart Melting point metals points metal alloys weights chart various lathe
When A Solid Is Converted Directly To A Gas, The Process Is Known As Sublimation.
Melting Is The Process By Which A Substance Changes From The Solid Phase To The Liquid Phase.
It Is The Opposite Of Freezing.
Melting Is The Conversion Of A Solid To A Liquid.
Related Post: