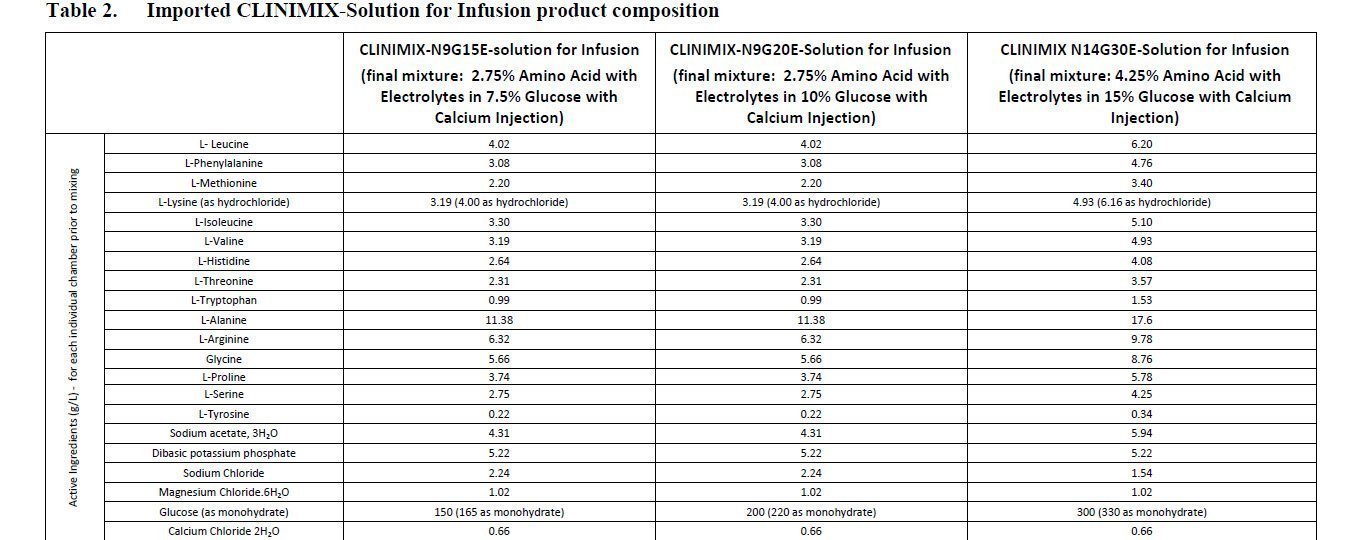
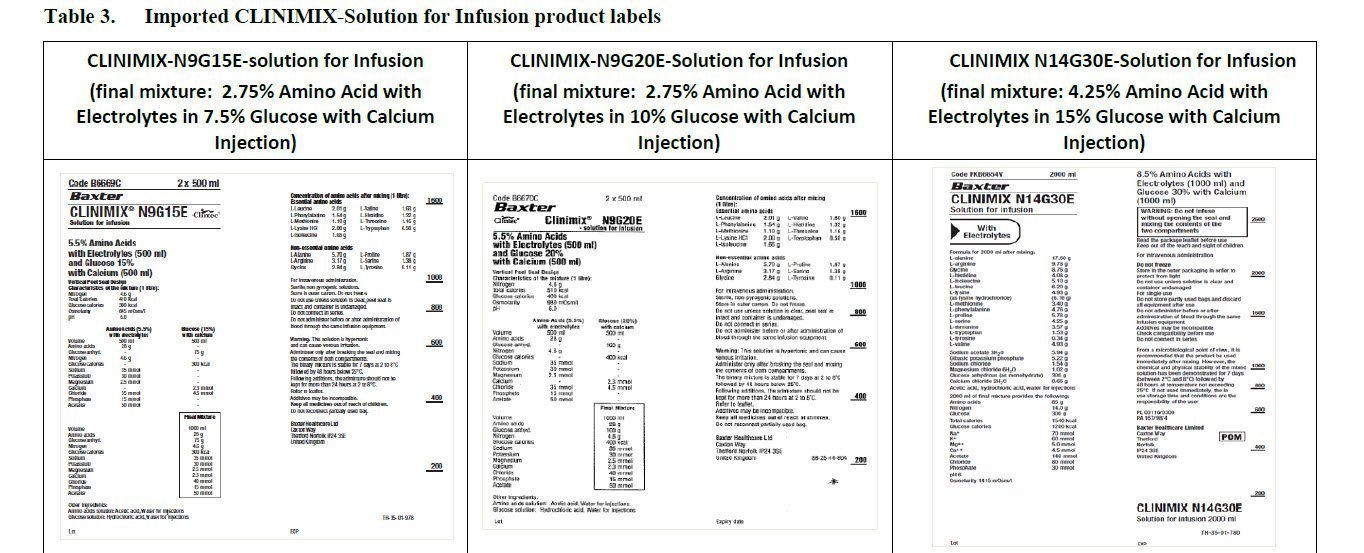
Clinimix Rate Chart
Clinimix Rate Chart - Please see indications and important risk information. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Clinimix is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. Detailed drug information for clinimix. Clinimix and clinimix e may be used to treat negative nitrogen balance in patients. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. A global leader in clinical nutrition, today announced the u.s. Food and drug administration (fda) approval of new formulations of clinimix (amino acids in dextrose). Clinimix is a preformulated total parenteral nutrition (tpn) intravenous formula. Includes common brand names, drug descriptions, warnings, side effects and dosing information. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. A global leader in clinical nutrition, today announced the u.s. It provides nutritional support to patients who are unable to obtain adequate nutrients orally due. Detailed drug information for clinimix. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Please see indications and important risk information. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Clinimix and clinimix e may be used to treat negative nitrogen balance in patients. Clinimix is a preformulated total parenteral nutrition (tpn) intravenous formula. Food and drug administration (fda) approval of new formulations of clinimix (amino acids in dextrose). Food and drug administration (fda) approval of new formulations of clinimix (amino acids in dextrose). Clinimix and clinimix e may be used to treat negative nitrogen balance in patients. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Please see indications and important. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. It provides nutritional support to patients who are. Detailed drug information for clinimix. Clinimix is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. Please see indications and important risk information. A global leader in clinical nutrition, today announced the u.s. Clinimix e is indicated as a source of calories, protein, and electrolytes. Clinimix is a preformulated total parenteral nutrition (tpn) intravenous formula. Includes common brand names, drug descriptions, warnings, side effects and dosing information. Clinimix is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. It provides nutritional support to patients who are unable to obtain adequate. It provides nutritional support to patients who are unable to obtain adequate nutrients orally due. Clinimix is a preformulated total parenteral nutrition (tpn) intravenous formula. A global leader in clinical nutrition, today announced the u.s. Clinimix is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or. Food and drug administration (fda) approval of new formulations of clinimix (amino acids in dextrose). Clinimix is a preformulated total parenteral nutrition (tpn) intravenous formula. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Clinimix e is indicated as a source of calories,. It provides nutritional support to patients who are unable to obtain adequate nutrients orally due. Clinimix is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. Includes common brand names, drug descriptions, warnings, side effects and dosing information. Please see indications and important risk information.. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Food and drug administration (fda) approval of new formulations of clinimix (amino acids in dextrose). Clinimix and clinimix e may be used to treat negative nitrogen balance in patients. Includes common brand names, drug. It provides nutritional support to patients who are unable to obtain adequate nutrients orally due. A global leader in clinical nutrition, today announced the u.s. Please see indications and important risk information. Clinimix and clinimix e may be used to treat negative nitrogen balance in patients. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients. Food and drug administration (fda) approval of new formulations of clinimix (amino acids in dextrose). Detailed drug information for clinimix. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. It provides nutritional support to patients who are unable to obtain adequate nutrients orally. Food and drug administration (fda) approval of new formulations of clinimix (amino acids in dextrose). Please see indications and important risk information. Clinimix is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. It provides nutritional support to patients who are unable to obtain adequate nutrients orally due. A global leader in clinical nutrition, today announced the u.s. Detailed drug information for clinimix. Includes common brand names, drug descriptions, warnings, side effects and dosing information. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,. Clinimix e is indicated as a source of calories, protein, and electrolytes for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient,.Clinimix FDA prescribing information, side effects and uses
Clinimix E FDA prescribing information, side effects and uses
Clinimix FDA prescribing information, side effects and uses
Clinimix FDA prescribing information, side effects and uses
Clinimix E FDA prescribing information, side effects and uses
Guillain Barre Syndrome
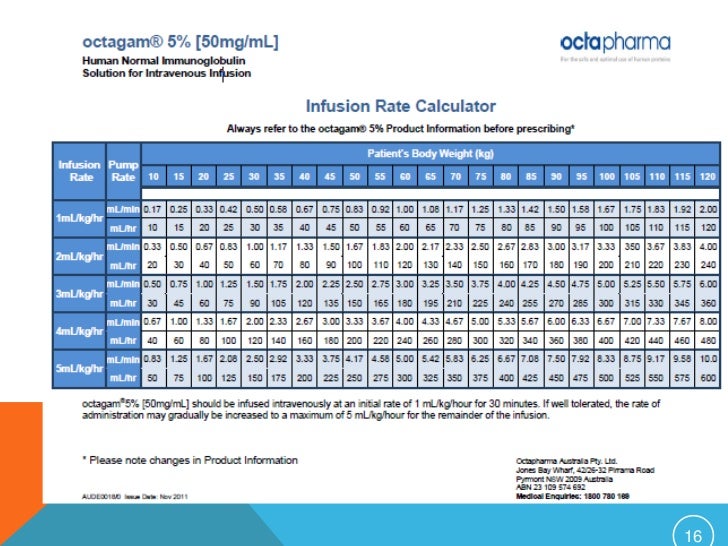
Continuous Infusion Rate Chart PDF
Clinimix FDA prescribing information, side effects and uses
Clinimix FDA prescribing information, side effects and uses
Clinimix E FDA prescribing information, side effects and uses
Clinimix Is A Preformulated Total Parenteral Nutrition (Tpn) Intravenous Formula.
Clinimix E Is Indicated As A Source Of Calories, Protein, And Electrolytes For Patients Requiring Parenteral Nutrition When Oral Or Enteral Nutrition Is Not Possible, Insufficient,.
Clinimix And Clinimix E May Be Used To Treat Negative Nitrogen Balance In Patients.
Related Post: